Japan Approves First iPS Cell-Derived Medical Treatments for Conditional Use
International News | World news Posted on 2026-03-08 07:07:37
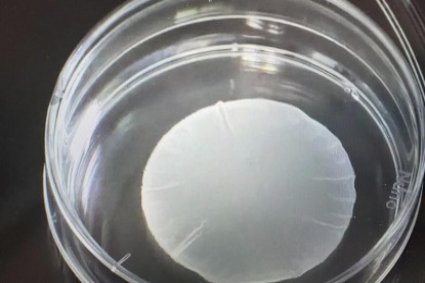
Japan has granted conditional approval for the production and sale of two regenerative medicine products developed from induced pluripotent stem (iPS) cells, marking what officials describe as the world’s first practical use of such technology.
The approval, announced Friday by the country’s health ministry, covers “ReHeart,” a cardiac muscle cell sheet developed by Tokyo-based startup Cuorips Inc., and “Amchepry,” a dopaminergic nerve cell therapy created by Sumitomo Pharma Co., Ltd. in Osaka.
Both treatments will become eligible for coverage under Japan’s public health insurance once the companies complete the required application process.
However, the approval is temporary. To obtain full authorization, the companies must gather additional clinical data on safety and effectiveness over the next seven years. Cuorips is expected to treat about 75 patients with ischemic cardiomyopathy using ReHeart, while Sumitomo Pharma plans to administer Amchepry to around 35 patients with Parkinson's disease.
According to Cuorips, ReHeart is a thin patch measuring about 4 to 5 centimeters in diameter and just 0.1 millimeter thick. Made from heart muscle cells derived from iPS cells—cells capable of developing into nearly any tissue—the patch is placed directly on the patient’s heart to help restore cardiac function. The company aims to begin selling the product in Japan this autumn and eventually expand into overseas markets.
Sumitomo Pharma’s Amchepry therapy involves transplanting between five million and ten million dopaminergic neural progenitor cells, also derived from iPS cells, into the brain to help improve motor function in Parkinson’s patients. The company plans post-marketing clinical trials at seven medical facilities, with the first transplant expected between October and December this year. Researchers will monitor patient progress for two years to evaluate the therapy’s safety and effectiveness.
Speaking at a news conference in Osaka, Sumitomo Pharma President and CEO Toru Kimura said the approval represents both an achievement and a new challenge. He described the development as a major milestone for bringing iPS cell technology into practical medical use.
Earlier this year, a ministry expert panel recommended granting the treatments time-limited approval while further research continues. Health Minister Kenichiro Ueno expressed hope that the new therapies could eventually benefit patients worldwide.
NPO News Team | PNA — PR
Share:









